Ion Exchange

Aeration
6 October 2020
Electrodeionization
8 October 2020
Ion exchange
The technology of ion exchange is often employed for aesthetic purposes in the form of water softeners to remove minerals, and it is also useful for the removal of inorganic chemicals, heavy metals and dissolved solids when used as water polishing technology. Fluoride, radium, and to some degree nitrate are all effectively treated with ion exchange resins. The removal of these contaminants is accomplished through adsorption of the contaminant ions onto a resin exchange medium. In this case, one ion is substituted for another ion on the charged surface of the medium, usually a synthetic plastic resin. This resin surface is designed as either cationic (negatively charged) or anionic (positively charged). The medium is saturated with the exchangeable ion before treatment operations begin. During exchange, contaminant ions replace regenerant ions because they are preferred by the medium. After the medium reaches equilibrium with the contaminant ions, the medium is regenerated with a suitable solution, such as sodium chloride, potassium chloride, caustic soda or acid solution. Ion exchange waste is highly concentrated and requires careful disposal.
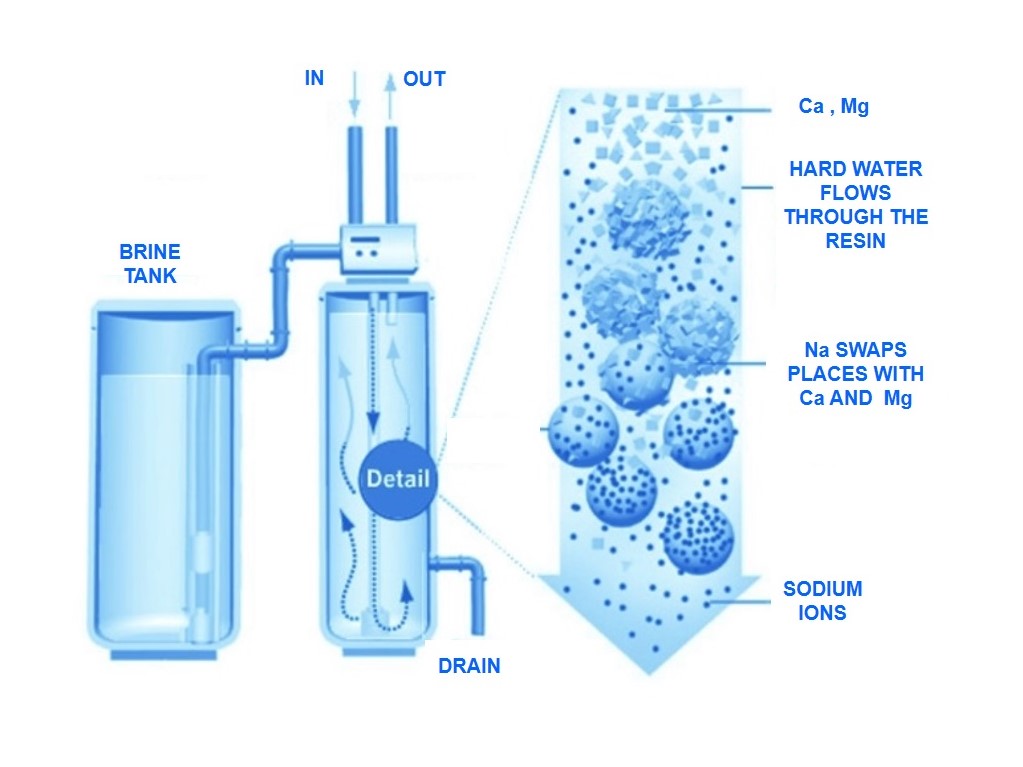
Water softening
Water softening is a process for the removal of calcium and magnesium from the water. These minerals are the main cause of hardness. With hard water, large quantities of soap are needed to produce a lather and a hard scale may form on cooking utensils or laundry basins. Hard, chalk-like formations may also coat the interior of pipes and water heaters.Most water softeners operate on the ion exchange process, which causes the replacement of calcium and magnesium ions with sodium ions. The exchange takes place when hard water comes into contact with an exchange medium, usually a synthetic resin or gel zeolite. These are insoluble granular materials that possess the property of ion exchange. After a period of time, the exchange material must be regenerated. The sodium ions are restored to the resin (or zeolite medium) by passing a salt (NaCl) or brine solution through the bed. However, this type of water softening process increases the sodium content of the water and may make it unsuitable for people on a low-sodium diet.
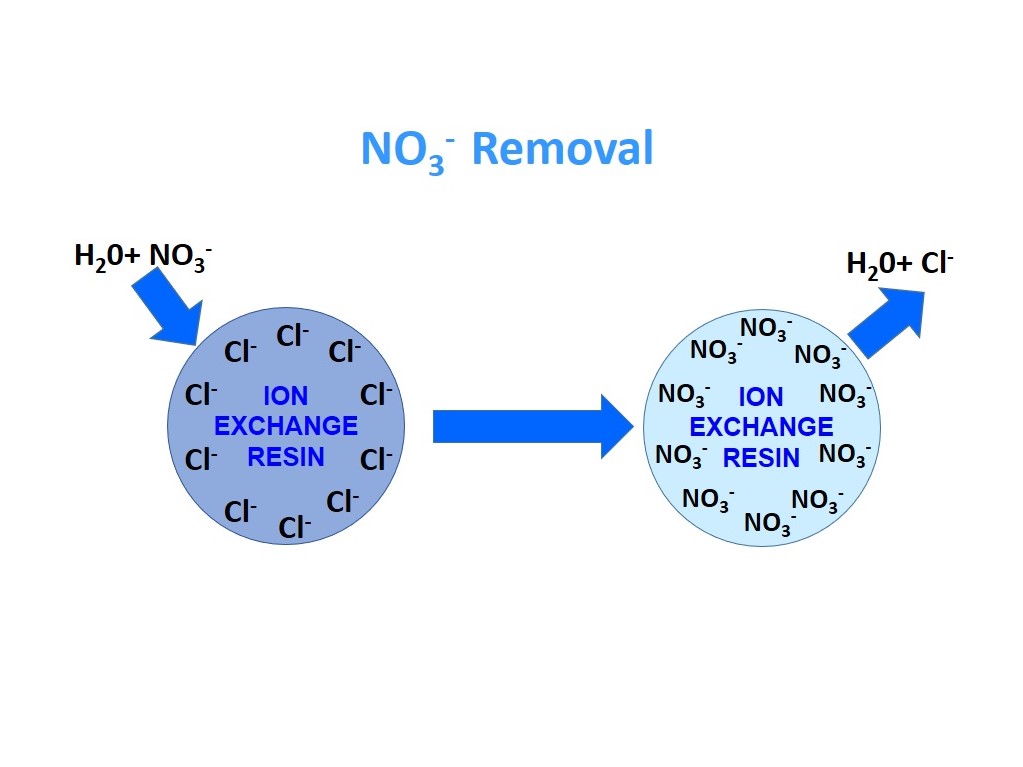
Nitrate (NO3) removal
Nitrate (NO3) is a compound that is formed when nitrogen combines with oxygen, sometimes high amounts of nitrate get into drinking water due to:fertilisers, animal wastes, particularly in areas of intensified farming, unreticulated sewage disposal systems industrial and food processing waste.Testing is the only way to detect nitrate, as it is tasteless, odorless and colorless. Nitrate levels do vary over the year often results are highest in spring (following rain and snow melt) and high nitrate levels have been found in late summer in areas where there is extensive irrigation.
Nitrate is a highly soluble and stable ion in water. Therefore, some techniques have been employed for efficient removal from water, such as adsorption, ion exchange, reverse osmosis and biological treatments. Some of these methods have high operational costs. The ion exchange process seems to be the most suitable for small water suppliers because of its simplicity, effectiveness and relatively low cost. The ion exchange process involves passage of nitrate loaded water through a resin bed containing strong base anion exchange resins on which nitrate ions are exchanged for chloride until the resin is exhausted.
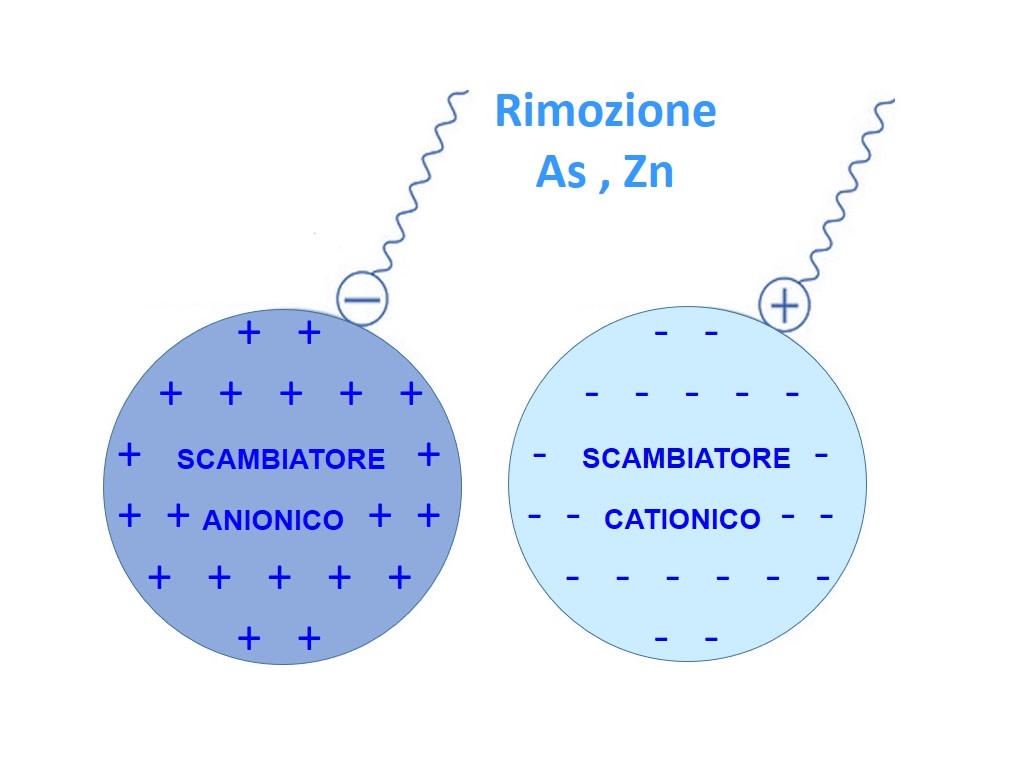
Metal ions removal: Arsenic (As), Zinc (Zn)
Ion exchange resins have good selectivity with respect to metal ions. It depends largely on the structure of the resin and specifically on the ionic radius and ionic charge density of the metal ions. An ion of higher charge density tends to displace an ion of smaller charge density. Trivalent ions are preferentially adsorbed, then divalent ions. Within ions of the same charge selectivity is governed by bond strength. This is determined by the electrovalent characteristic of the metal ion. Metal ions which are more electrovalent bind more strongly and are preferentially adsorbed. For example, copper is more electropositive than zinc; as a result, from an effluent containing dissolved copper and zinc, cupric ions are preferentially adsorbed. Arsenic is a toxic substance with varying degrees of regulation. By the technology of ion exchange it is possible by using anion resin to remove pentavalent and trivalent arsenic from water. Pre-oxidation is recommended for converting trivalent arsenic to the pentavalent form before removal by Ion Exchange Resins. Any excess of oxidant should be removed before contact with the resin.

Boron removal
Boron is a natural element that is prevalent in ground water from areas with volcanic geology, present in the sea water and is used in commercial products such as detergents. Mildly toxic to humans but frequently very toxic to plants, especially to citrus varieties, boron is regulated in most regions of the world. Depending on the water salinity, boron concentration and temperature, two main processes are used to produce drinking water below 0.5 mg/L of Boron:• 2-pass Reverse Osmosis design:
2nd-Pass Reverse Osmosis technology design with caustic soda addition to raise pH around 9.5. Some of the first pass permeate can be by-passed in order to keep some minerals in the water.
• Reverse Osmosis technology + Ion Exchange design:
Selective Boron Ion Exchange Resin with or without by-pass may be engaged for Boron removal as a post treatment of first pass permeate, depending on the residual boron concentration needed. The selective resin must be on-site regenerated with caustic soda and hydrochloric or sulphuric acid. A double column system is often required to ensure a continuous production.

