Filtration Technology

Microfiltration – Ultrafiltration – Nanofiltration
3 October 2020
Disinfection
5 October 2020
Filtration technologies
Filtration is the process of removing suspended solids from water as the water passes through a porous bed of materials
Natural filtration removes most suspended matter from ground water as the water passes through porous layers of soil into aquifers (water-bearing layers under the ground).Additionally, surface waters, are subjected to runoff and are inherently vulnerable to other sources of contamination such as sewage overflows, industrial discharges, fuel or chemical spills. Surface waters must therefore be filtered by some method in addition to being disinfected prior to use.
The conventional Conventional Filtration, also known as rapid-sand filtration, this is the most prevalent type of water treatment technology used today. This filtration technique employs a combination of physical and chemical processes in order to achieve maximum effectiveness, as follows:

Coagulation and flocculation
Chemicals are added to the water in order to improve the subsequent treatment process. These may include pH adjusters and coagulants. Coagulants are chemicals, such as alum, that neutralize positive or negative charges on small particles, allowing them to stick together and to form larger particles that are more easily removed by sedimentation (settling) or filtration. A variety of devices, such as baffles, static mixers, impellers, and in-line sprays can be used to mix the water and distribute the chemicals evenly.
Flocculation: In this process, which follows the rapid mixing, the chemically treated water is sent into a basin where the suspended particles can collide, agglomerate (stick together), and form heavier particles called “flocs”. Gentle agitation of the water and appropriate detention times (the length of time water remains in the basin) help facilitate this process.
The factors, which can promote the coagulation-flocculation, are the velocity gradient, the time, and the pH. The time and the velocity gradient are important to increase the probability of the particles to come together. Moreover the pH is a prominent factor in the removal of colloids.

Sand filter
Sand filtration represents a technology that has been used for years in various areas. and only recently has it been re-examined as a cost-effective, efficient, and relatively simple system for water filtration. In a slow sand filter, raw water passes by gravity through a bed of sand, about 1 m deep, which is supported underneath by a layer of gravel. Filtered water is collected by an under drain system which is placed within or underneath the gravel. A thin layer of biologically active microorganisms’ forms (if there is no a pre-chlorination treatment) in the on top of and throughout the sand bed. These organisms filter out sediment and kill harmful bacteria. After a period of time, the filter bed will begin to clog due to trapped particulate matter. A sand filter bed with a relatively uniform grain size can provide effective filtration throughout its depth. If the grain-size gradation is too great, effective filtering is confined to the upper few inches of sand. This results because the finest sand grains accumulate on the top of the bed during stratification after backwashing. The problem of surface plugging of sand filters led to development of dual-media filters.
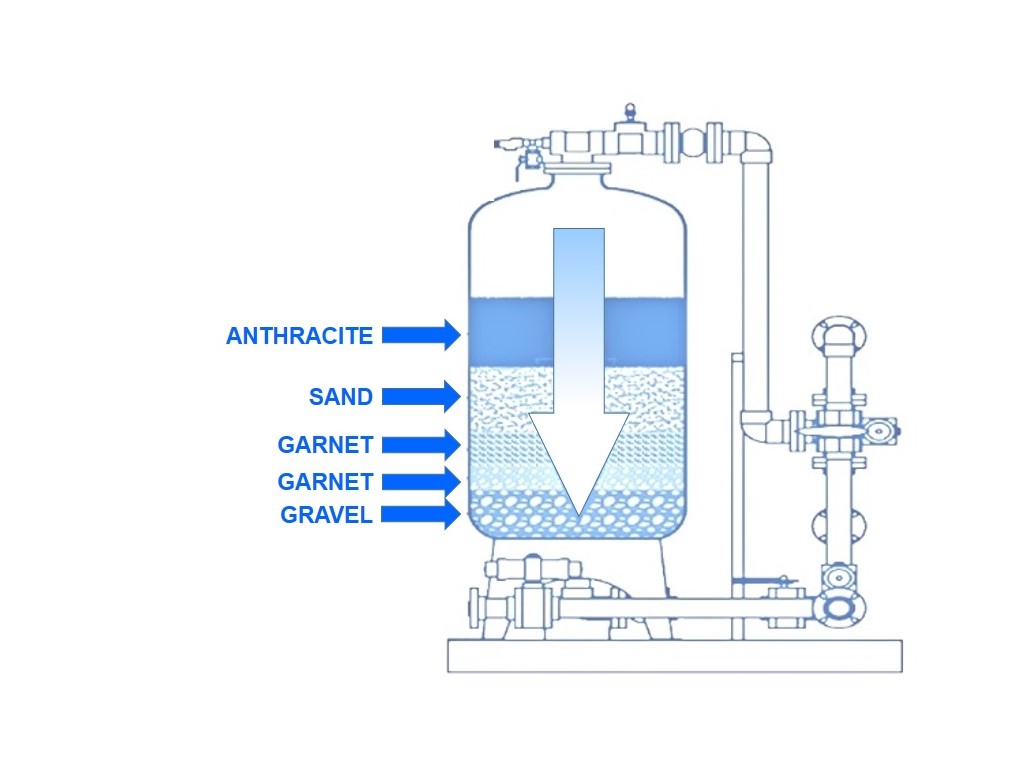
Dual media – Multimedia fitration
A dual-media filter consists of a sand [specific gravity (sg), 2.65] layer topped with a bed of anthracite coal medium (1.4 - 1.6 sg). The coarser anthracite top media layer pores about 20% larger than the sand medium. These openings are capable of adsorbing and trapping particles so that floc carried over in clarified water does not accumulate prematurely on the filter surface and plug the sand filter. The Multimedia Filters MMF, have more than one medium, they may be open gravity filters or pressure filters. In water treatment, they have become more popular in recent years. Dual-media filter beds usually employ anthracite and sand; however, other materials have been used, such as activated carbon and sand. Multimedia filter beds generally use anthracite, sand, and garnet. However, other materials have been used, such as activated carbon, sand, and garnet. Also, dual- and multimedia filters using ion exchange resins as one of the media have been tried. In some of these filters, the media may have additional characteristics other than removing particles. For example, activated carbon removes dissolved organic substances.
The process of multimedia filtration produces high quality, filtered water at much faster flow rates than traditional sand filtration.

Filtration with activated carbon
Along with aeration, granular activated carbon (GAC) and powdered activated carbon (PAC) are suitable treatments for removal of organic contaminants such as VOCs, solvents, PCBs, herbicides and pesticides. Activated carbon is carbon that has been exposed to very high temperature, creating a vast network of pores with a very large internal surface area; one gram of activated carbon has a surface area equivalent to that of a football field. It removes contaminants by adsorption, a process in which dissolved contaminants adhere to the surface of the carbon particles.. After a period of a few months or years, depending on the concentration of the contaminants, the surface of the pores in the GAC can no longer adsorb contaminants and the carbon must be replaced.Powdered activated carbon consists of finely ground particles and exhibits the same adsorptive properties as the granular form. PAC is normally applied to the water in a slurry and then filtered out. The addition of PAC can improve the organic removal effectiveness of conventional treatment processes and also remove tastes and odors.

Iron/Manganese removal filter
The filters for the removal of iron and manganese from water using sand of different granulometries and pyrolusite (MnO2) or other media. After the oxidation step (with or without a detention or settling tank), the source water is filtered through a filter media in either a pressure vessel or a gravity filter. In the presence of dissolved oxygen, the pyrolusite exerts a strong catalytic oxidation action on the Fe and Mn, which precipitates and is trapped in the filter bed. Later, tits eliminated by backwash. It does not need to be regenerated, and is not consumed during the process, thus having a very long shelf life. The filtration media in these systems may consist of sand, sand and coal anthracite (dual media), or propri¬etary/patented products, such as Pyrolusite - Pyrolox, Filox-R, Birm, and manganese greensand. Some media such as manganese greensand, have the ability to both oxidize and filter iron and manganese effectively and at the same time. Manganese greensand, pyrolusite, Birm, or any media coated with manganese dioxide has the capacity to oxidize iron and manganese and filter the insoluble precipitates with the filter bed. These media also have some, but limited, capacity for As(III) oxidation and arsenic adsorption.

Pyrolusite
Pyrolusite is the common name for naturally occurring manganese dioxide. It is a mined ore consisting of 40 to 85% manganese dioxide by weight. The various configurations of pyrolusite provide extensive surface sites available for oxidation of soluble iron and manganese. Removal rates of iron in excess of 20 mg/L are achievable.Pyrolusite may be used in the following two ways: (1) Mixing with sand, typically at 10¬-50% by volume, to combine a filtering media with the oxidizing properties of pyrolusite; (2) Installing 100% pyrolusite in a suitably graded filter to provide oxidation and filtration.
Maximum hydraulic loading rates of 9,0-12,50 m³/m² should be the basis of design for a pressure vessel. No chemical regeneration is required. Backwash is critical for proper operation. Attrition during backwash can be a benefit as it exposes more surface sites for oxidation of soluble iron and manganese. The density of pyrolusite is in the range of 120 lb/ft³, requiring a backwash rate of 50-60 m³/m² to fluidize the bed, scrub the media, and redistribute the media throughout the bed. Air scour and backwashing are recommended in simultaneous mode.

Birm
Birm is an acronym that stands for the “Burgess Iron Removal Method”.. Birm has the capacity to oxidize iron, but is not very effective at oxidizing As(III) to As(V). Birm is produced by impregnating manganous salts to near saturation on aluminum silicate sand, a base material. The manganous ions then are oxidized to a solid form of manganese oxide with potassium perman¬ganate. This process is similar to that used to manufac¬ture manganese greensand. To be effective, it must be used in water with a pH range of 6.8-9.0.Alkalinity should be greater than two times the combined sulfate and chloride concentration. Injection of com¬pressed air ahead of the media to maintain a dissolved oxygen content of at least 15% of the iron content may be required, especially for source water with iron at concentrations greater than 3 mg/l.¬ The dissolved oxygen oxidizes iron with Birm media serving as a catalyst that enhances the reaction between dissolved oxygen and dissolved iron and manganese in the water. Further, formed ferric hydroxide attracts oxidized arsenic, which then is captured in the filter bed.

Manganese greensand
Another media that converts soluble forms of iron and manganese to insoluble forms that can then be filtered is manganese greensand. Manganese greensand has been used for several decades and is formed from processed glauconite sand. The glauconite is synthetically coated with a thin layer of manganese dioxide, which gives the dark sand a definite green color and thus its name. Limitations for manganese greensand include a maximum limit of 5 mg/L of hydrogen sulfide removal and 15 mg/L for iron removal; also, water pH should be in the range of 6.2-8.5.The combination of a strong oxidant and manganese greensand filtration media for iron removal is commonly referred to as the “Manganese Greensand Process.” Either potassium permanganate or chlorine can be used to effectively regenerate manganese greensand filters. However, if chlorine is used alone, it may be necessary to periodically regenerate the manganese greensand using potassium permanganate by a batch process in order to maintain optimum effectiveness of the media. Prechlorination is often recommended if iron levels are significantly greater than 1 mg/L in order to reduce the need for the more expensive potassium permanganate.

